
Contact person: tony.wang
Address: No 65 Science Avenue, HI-TECH Zone, Zhengzhou, China
Views: 11 Author: Site Editor Publish Time: 2021-12-16 Origin: Site
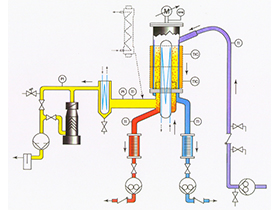
Molecular distillation is also called short-path distillation. It is a relatively new liquid-liquid separation technology that has not been widely used in industrial production. Its application can solve a large number of problems that cannot be solved by conventional distillation technology. Molecular distillation is a distillation method that operates under high vacuum. At this time, the mean free path of vapor molecules is larger than the distance between the evaporation surface and the condensation surface, so that the difference in the evaporation rate of each component in the liquid can be measured. Use it to separate liquid mixtures. Molecular evaporator, degassing system, feeding system, heating system, cooling vacuum system and control system are the main components of a complete molecular distillation apparatus. The molecular evaporator is the core part of the molecular distillation device.
The short path distillation apparatus is quite suitable for molecular distillation, the molecular flow from the heating surface directly to the surface of the condenser. Molecular distillation has the following four processes:
1. Molecules diffuse from the liquid phase to the evaporation surface
Generally, the diffusion rate in the liquid phase is the main factor controlling the molecular distillation rate, so the thickness of the liquid layer should be reduced as much as possible and the flow of the liquid layer should be strengthened.
2. Free evaporation of molecules on the surface of the liquid layer
As the temperature rises, the evaporation rate continues to rise. However, as the temperature rises, the separation factor sometimes decreases. Therefore, the thermal stability of the processed material must be used as a prerequisite to select an economical and reasonable distillation temperature.
3. Molecules fly from the evaporation surface to the condensation surface
When the vapor molecules fly from the evaporation surface to the condensation surface, they may collide with each other, and may also collide with air molecules remaining between the two surfaces. Because evaporating molecules are much heavier than air molecules and most of them move in the same direction, their collisions will not have a large impact on the direction of flight and the speed of evaporation. However, the chaotic thermal motion state of residual gas molecules appears between the two sides, so the main factor that affects the direction of flight and the evaporation speed is the number of residual gas molecules.
4, molecules condense on the condensation surface
It is only necessary to make the temperature difference between the hot and cold sides sufficient (usually in the range of 70-100 degrees Celsius), and the form of the condensing surface is reasonable and smooth, then it is considered that the condensing step can be completed in an instant, so it is very important to have a reasonable condenser The choice of form.
Contact person: tony.wang
Email:tony.wang@mingyint.com
kevin.wang@mingyint.com
Address: Room 1002 10/F, Easey, Commercial Building 253-261 Hennessy Road WanChai, HK.
Address: No.65 Science Avenue,HI-TECH Zone,Zhengzhou,China.